Topic #3: Membraneless (Photo)electrochemical cells and reactors
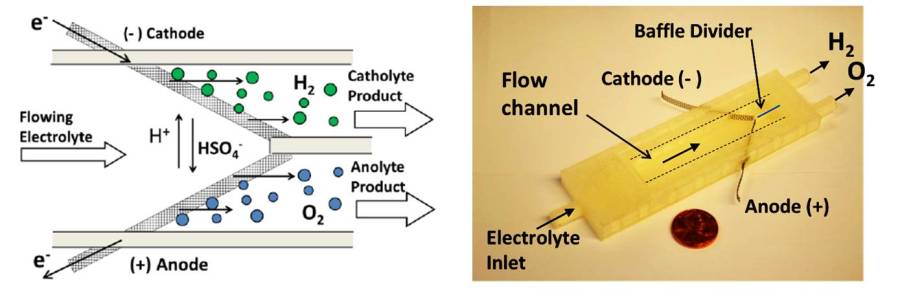
Of particular interest in our group are novel membraneless PEC and electrochemical reactors, which offer exciting opportunities to decrease capital costs through their simplicity. Additionally, membrane-free devices have potential advantages in terms of durability and electrolyte flexibility that can enable electrolyzers to be used in new operating regimes and/or applications. Two key aspects of of our device-level research are (i) Additive manufacturing (3D-printing), which has proven to be invaluable for accelerating the development of electrochemical and photoelectrochemical test cells and reactors, and (ii.) Incorporation of windows and electrochemical sensors into these devices. The former features enable in situ studies of the fluid dynamics and bubble dynamics within these devices using high speed video (HSV). These measurements are carried out in close collaboration with electrochemical engineering (modeling) and CFD experts to validate models and develop design guidelines for these devices.
High speed video of H2 and O2 gas bubble evolution from Platinized Ti mesh operating in 0.5 M H2SO4 at 150 mA/cm^2. By coating the Pt electrocatalyst only on the outer surfaces of the mesh electrode (left video), gas bubbles can be collected with product purities as high as 99% while the holes in the porous mesh electrode still allow for ions to transport between the anode and cathode. Complete details can be found in reference [1]. Videos taken by Jack Davis. Video formatting credited to a Columbia University press release.
Select Publications in the Research Area:
Review or Perspective Articles
- D.V. Esposito, “Membraneless Electrolyzers for Low-Cost Hydrogen Production in a Renewable Energy Future”. Joule, 1, 1-8, 2017. (Perspective) Download here.
- Mark T. Spitler, et al.. “Practical Challenges in the Development of Photoelectrochemical Solar Fuels Production”. Sustainable Energy & Fuels (2020). Doi: 10.1039/C9SE00869A. Download here.
Original Research Articles
- G.D. O’Neil, C. Christian, D.E. Brown, D.V. Esposito, “Hydrogen Production with a Simple and Scalable Membraneless Electrolyzer”. Journal of the Electrochemical Society, 162, F3012-F3019, 2016. Download here (Open Access).
- J.T. Davis, D.V. Esposito, “Limiting Photocurrent Analysis of a Wide Channel Photoelectrochemical Flow Reactor”, Journal of Physics D: Applied Physics., vol. 50, 8, 11 pp, 2017. (Special Issue on Solar Fuels). Available for download here.
- O.O. Talabi, A.E. Dorfi, G.D. O’Neil, D.V. Esposito, “Membraneless Electrolyzers for the Simultaneous Production of Acid and Base”. Chemical Communications, 53, 8006-8009 2017. Part of the 2017 Emerging Investigators Issue. Download here.
- J.T. Davis, J. Qi, X. Fan, J. Bui, D.V. Esposito, “Floating Membraneless PV-Electrolyzer Based on Buoyancy-Driven Product Separation”, International Journal of Hydrogen Energy, vol. 43 (3), pp 1224–1238, 2018. Download here. Highlighted in a Columbia University press release.
- J. Davis, D. Brown, X. Pang, D.V. Esposito, “High Speed Video Investigation of Bubble Dynamics and Current Density Distributions in Membraneless Electrolyzers”. Journal of the Electrochemical Society, vol. 166 (4), pp F312-F321, 2019. Download here.
- J.C. Bui, J.T. Davis, D.V. Esposito, “3D Printed Clickable Membraneless Electrode Assemblies for Water Electrolysis”. Sustainable Energy & Fuels , vol. 4, 213-225, 2020. Download here.
- X. Pang, S. Das, J. Davis, A. Harvey, D.V. Esposito, “Framework for evaluating the performance limits of membraneless electrolyzers”. Energy & Environmental Science, Advance Article, doi: 10.1039/D0EE02268C . *Note* This is an open access article that is freely available for download through this link: Download here.
- N. Mahmud, D.V. Fraga Alvarez, M.H. Ibrahim, M.H. El-Naas, D.V. Esposito, “Magnesium recovery from desalination reject brine as pretreatment for membraneless electrolysis”. Desalination, vol. 525, 115489, 2022. Download here.
- X. Pang, S. Verma, G. Liu, D. V. Esposito, “Membrane-free Electrochemical CO2 Conversion using Serially-Connected Porous Flow-Through Electrodes”. Joule, 2022, 6 (12), P2745-2761. Download here.